This year around, we had a really great group of students working in the Course on Laboratory Excercises in Radiochemistry. That’s the reason we decided that, instead of doing the regular Neutron Activation Analysis example where we determine the elemental composition of ground coffee, we would go for something slightly more fun:

With much skill, the students collected a 5 ml sample of Espresso while it was coming out of the machine! 
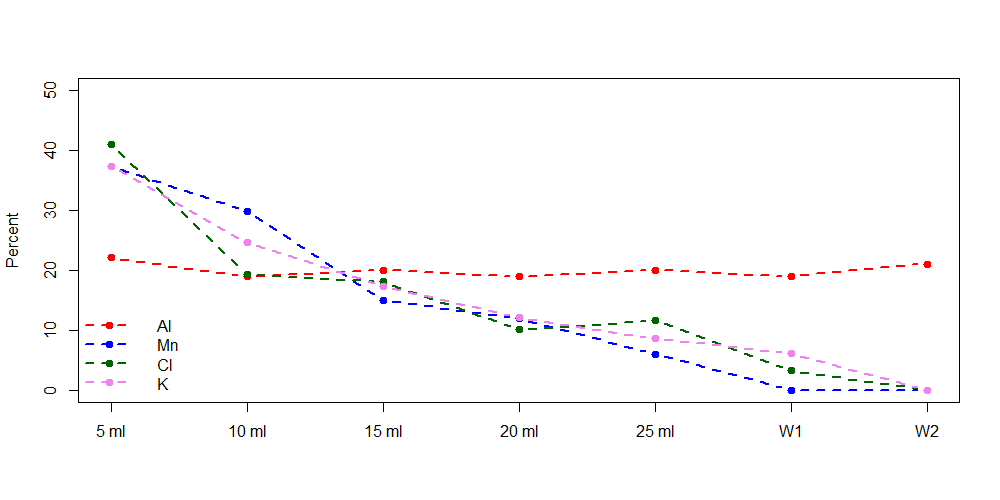
Two more samples (6 and 7) were taken, one from the machine without any coffe inside and one from the tab we usually use to fill the water container of the machine. The first sample is a reference sample we mixed from KMnO4, NaCl, KNO3, Al(NO3)3.9H2O and Mg(CH3COO)2.4H2O. From each sample we then took a 1 ml aliquot and irradiated them sequentially with our slow pneumatic transport system for 60 s each at an approximate neutron flux density of cm-2s-1. Measurements were done after a decay time of 3 minutes for 1 minute each. From the resulting spectra, the students were able to calculate the elemental composition of each sample (for the elements Al, Mn, Cl and K). We also tried to look for Na, but the Na content was below the background in most samples.
The results show that within the first 10 ml at least half of the elemental content that is not in the water already (last two samples) is extracted out of the coffee. So for all those enthusiats who like their coffee short, here’s another good reason!